Battery Products and Power Solutions

 Battery Products
Battery Products
Portable power solutions from design to delivery for over 50 years
Custom Power has been an industry leader in the design and assembly of battery solutions since 1965. We supply battery pack assemblies in all chemistries across a diverse range of applications, from military equipment and medical instruments to emergency lighting systems and tracking devices. We give our customers the competitive edge they need to succeed by optimizing battery performance, cost, size and working life.
We are one of the largest authorized custom battery assemblers in the United States, providing solutions to some of the world's most recognized companies. Our engineering team has decades of combined experience developing customized solutions to meet a broad range of needs. We stand out in the market for our expertise in battery management systems, circuit design and complex battery pack assemblies.
 Types of Battery Products We Offer
Types of Battery Products We Offer
Custom Power offers a comprehensive range of products and custom designs for lithium-ion, lithium polymer, primary lithium, nickel-metal hydride (NiMH), nickel-cadmium (NiCd) and alkaline batteries. Explore some of the specific product types we supply.
Custom Battery Packs
Our engineers can create custom packs with primary or rechargeable cells using various chemistry options.
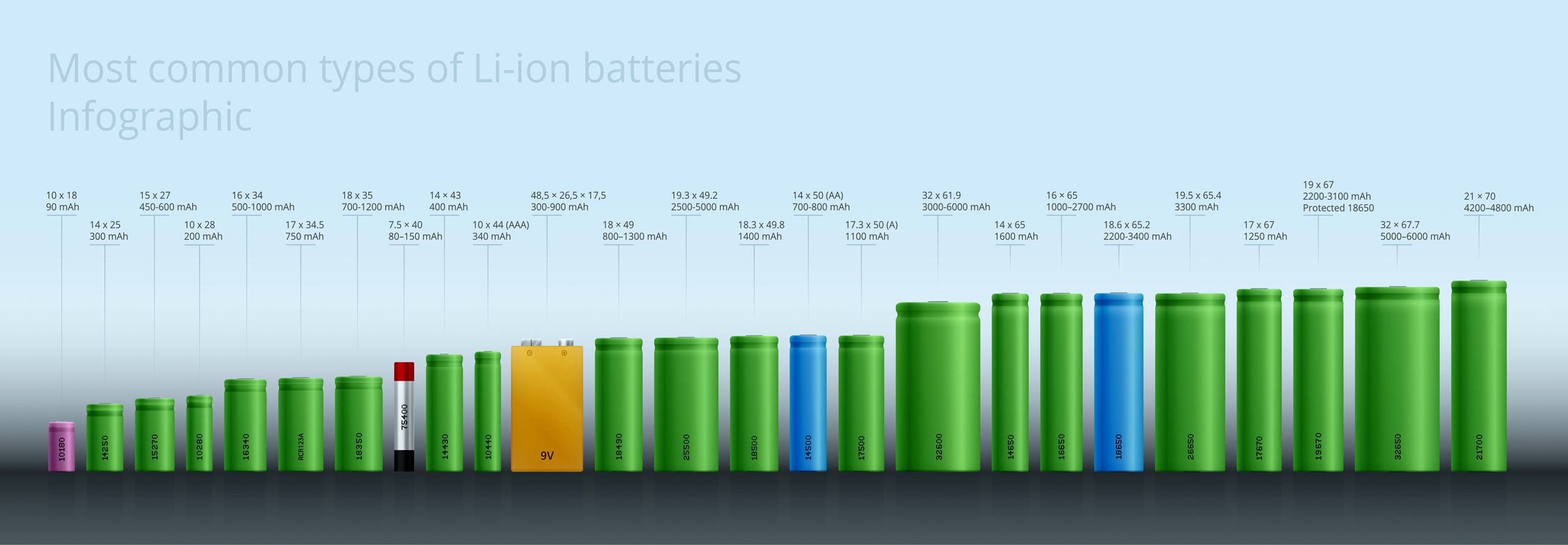
Standard Batteries
Our standard battery cell and product selection are one of the most comprehensive on the market.
Battery Chargers
Regardless of the type or size of the battery you use, we can design a reliable, high-performance charger to fit your needs.
Battery Suppliers
Custom Power partners with many top-tier battery suppliers to help create solutions optimized for your application.
 Custom Power Delivers Reliable Energy Solutions
Custom Power Delivers Reliable Energy Solutions
When you partner with Custom Power, you will work with some of the most knowledgeable and skilled engineers in the battery industry.
Several factors make us a global leader:
 Frequent Applications
Frequent Applications
At Custom Power, we supply engineered battery solutions to address the needs of many applications, including:
 Contact an Expert
Contact an Expert
Whether you're looking for an off-the-shelf product or a customized battery pack unique to your application, Custom Power has an option for your needs. Contact us online today to request a quote or additional product information.